Isotope effect on structural
transitions in Y0.9Gd0.1Fe2(HzD1-z)4.2 compounds
Chem.
Met. Alloys 6
(2013) 130-143
https://doi.org/10.30970/cma6.0249
Valérie Paul-Boncour, Svetlana Voyshnis, Karine
Provost, Jean-Claude Crivello
Y0.9Gd0.1Fe2(HzD1-z)4.2 compounds
crystallize in a monoclinic structure at room temperature, with an increase of
the cell parameters versus the H
content. These compounds undergo a ferro-antiferromagnetic
first-order transition, the transition temperature of which increases from 98
to 144 K for z going from 0 to 1, due
to a strong magnetovolumic effect. Above room
temperature (290-340 K), they display an order-disorder (O-D) transition from
monoclinic towards a cubic structure, which has been studied by DSC and XRD versus
temperature. This transition occurs via
the presence of an intermediate phase, the structure of which depends on the H
content. For z = 0 and 0.5 the intermediate phase
is monoclinic, whereas an orthorhombic phase is observed for z = 0.75 and 1. In addition, for the H-rich compounds the
orthorhombic phase disappears at a much lower temperature upon cooling than it
appears upon heating. DFT band structure calculations
for YFe2Hx
compounds showed that for 4 < x < 4.5, although the monoclinic phase is the more stable one,
the energy of formation of the orthorhombic phase is only 0.1 kJ higher. For 4.5 < x ≤ 5
the orthorhombic phase becomes more stable. The sensitivity of the O-D
transition to the H/D content could be related to a volume effect. At higher
temperatures (T > 400 K), the
thermal desorption studied by TGA
shows a multipeak behavior that is not sensitive to
the (H, D) isotope effect.
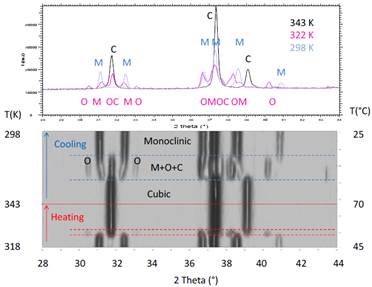
Diffraction patterns versus temperature of Y0.9Gd0.1Fe2(H0.75D0.25)4.1
upon heating and cooling.
Keywords
Laves
phases / Isotope effect / X-ray diffraction / Order-disorder transition / DFT calculation