Synthesis and catalytic properties of Mg, Co(II),
Zn phosphate solid solutions
Chem.
Met. Alloys 6
(2013) 70-74
https://doi.org/10.30970/cma6.0242
N.M. ANTRAPTSEVA, N.V. SOLOD, L.B. KOVAL
Dihydrogenphosphates with the general formula Zn1-хСоx(H2РO4)2·2Н2О (0 < х < 1)
and phosphates of composition Co3-хMgх(PO4)2·8Н2О (0 < х ≤ 1)
were synthesized. The former crystallize
in the monoclinic system, space group Р21/n
(Z = 2), with the formation of a continuous substitutional solid solution. The latter form a limited
solid solution. Their catalytic properties were investigated. The effectiveness
of their action as catalysts for alcohol dehydration and methane oxidation was
shown.
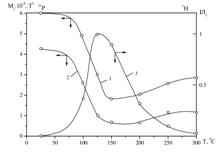
Interrelation of results of heat
treatment of Zn0.5Со0.5(H2PO4)2·2H2O
at different temperatures as obtained by proton NMR and 31Р NMR (1 –
width and 2 – second moment of lines in the 31Р NMR
spectra; 3 – intensity of the narrow component in the 1H NMR spectra)
Keywords
Phosphate / Solid solution / Synthesis /
Catalyst